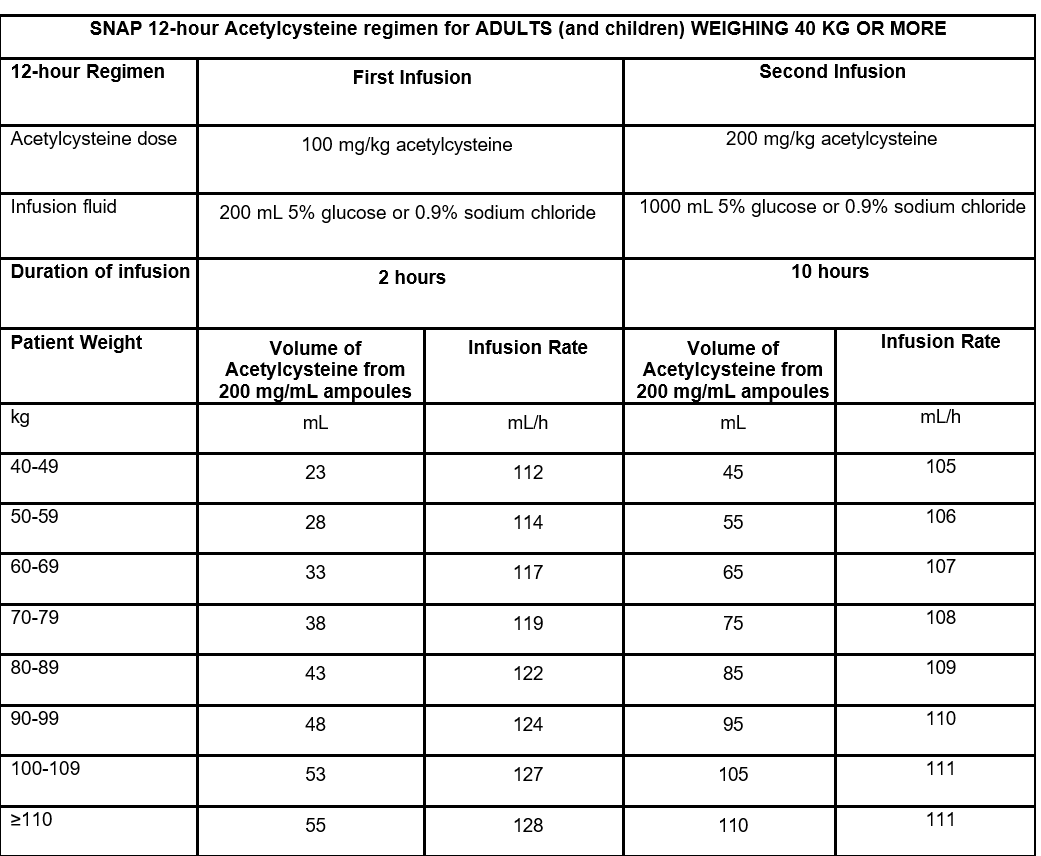
If there is any doubt as to the total dose ingested or concern regarding patient symptoms then NAC should be continued (3rd bag) at the dose and infusion rate used in the second bag (10 hour).
Bloods: if available re-check ALT, U&Es, PT (or INR), paracetamol concentration. Bloods should be taken after the 2nd bag has finished. Results from bloods taken whilst the infusion is running may underestimate paracetamol concentrations or overestimate the INR.
Requirement for 3rd infusion - WITHOUT ALT and / or paracetamol concentrations:
If ALT and paracetamol concentrations are not available at the end of the 12-hour regimen then NAC maybe stopped if:
- less than 150 mg/kg paracetamol has been ingested
AND
- the patient has no symptoms of hepatotoxicity or acute kidney injury.
AND
- available blood results e.g VBG,FBC, U&E, PT are normal.
Patients should then be observed for a further 12 hours and NAC restarted if symptoms develop.
If these criteria are not met, NAC should be continued at the dose and infusion rate used in the second bag (10 hour).
Requirement for 3rd infusion when ALT and paracetamol concentrations available:
NAC should be continued if ANY of the following criteria are met (continue NAC at the dose and infusion rate used in the second bag (10 hour):
- The ALT is above the upper limit of normal range
OR
- The ALT has doubled or more from admission (even within the normal range)
OR
- The paracetamol concentration is greater than 10 mg/L
Management of Patients receiving 3rd bag or more.
In the deployed setting any patient with features of hepato-toxicity and/or renal toxicity should receive additional infusions (bags) of NAC at the same dose and rate as the 2nd (10-hour) bag, until they can be evacuated to a location with appropriate laboratory capabilities.
Bloods: Recheck ALT, U&Es, INR at the end of each 10-hour bag. There is no need for paracetamol concentrations unless this was available and raised > 10 mg/L after the 2nd bag.
End of 3rd Bag (22 hours) - criteria to continue NAC if ANY of the following are present:
- ALT more than two times the upper level of normal
OR
- ALT above upper level of normal AND increased from previous result AND more than doubled since admission
OR
- PT is above upper level of normal AND increased from previous value AND the ALT is above the upper level of normal.
End of 4th or subsequent bags – criteria to continue NAC
After the 4th bag the decision for further NAC requirement is made on the INR (PT) alone. NAC should be continued until:
- PT is normal (or INR is less than 1.3)
OR
- PT is falling towards normal on two consecutive blood tests and is less than (INR 3.0)
Patients who do not meet the criteria for further NAC can be discharged.