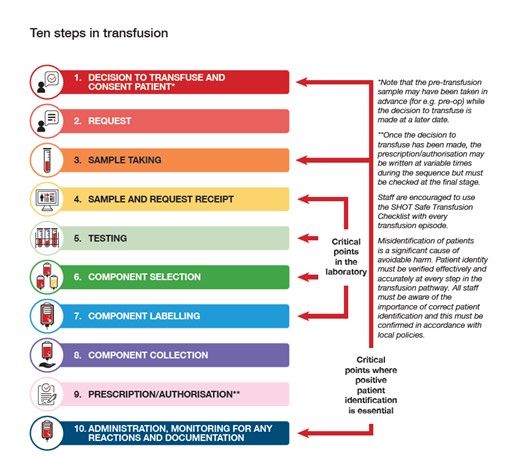
Positive identification MUST occur at every step of the transfusion process: blood sampling, component selection, component administration and post transfusion observation.
Positive identification - Patient must state their full name and date of birth. Clinical staff MUST confirm details match those on wrist band / identifier. In cases where the patient has a reduced level of consciousness, confirmation should be performed with a second person reading details direct from wrist band.
Blood selection and administration requires a minimum of 4 identifiers to coordinate:
Last-name, first-name, date of birth and unique identification number1 are required for provision of group specific products (providing sample testing to confirm patients’ blood group is available). Where possible the patient sex at birth must be stipulated.
Samples collected for grouping or crossmatching must also have phlebotomists initials included.
For casualties with unknown identity, the local unidentified patient protocol should be followed. This must involve unique identifiers, such as those detailed on casualty triage cards. Minimum data set is for patients with unknown identity are:
- Unique Identifier e.g. trauma number
- Service number
- Sex
- Time and date of sample collection
For casualties receiving universal blood components (Group O Red Cell concentrate /Whole blood, Group A Platelets and Group AB/A FFP) the above principles must still be followed to ensure patient safety and retain confidence in traceability documentation.
1Regimental number, ZAP number or Unique trauma number generated in medical facility