
Major Haemorrhage Guideline
Objectives
- To ensure a safe, appropriate, and efficient transfusion service to all patients on Operations
- To give up to date and agreed standardisation on triggering a major haemorrhage response on Operations.
- To supplement current resuscitation guidelines and specialist policies.
- To provide an agreed protocol for the management of major haemorrhage, that guides all involved parties and minimises transfusion delays.
- To consider alternatives to transfusion wherever possible.
The recommendations are based on perceived current best practice that is subject to change.
Scope
To prevent the 'diamond of death' (acidosis, coagulopathy, hypocalcaemia and hypothermia) associated with major haemorrhage, early recognition of major blood loss and intervention is vital. Transfusion of blood components within minutes of recognising a major haemorrhage is crucial to improving patient outcomes.
The management of a major haemorrhage requires a multidisciplinary approach, with transfusion being one component in its management. This major haemorrhage policy is intended to supplement current resuscitation guidelines and specialist policies.
In the event of a major haemorrhage, it is necessary to balance the need to access blood components rapidly with the rigorous procedures aimed at avoiding inappropriate use of blood components, wastage and to ensure patient safety.
Audience
Deployed Primary and Secondary clinicians
Deployed Nursing staff
Biomedical Scientists
Allied Health Care Professionals
Initial Assessment & Management
Major haemorrhage
Most definitions of massive haemorrhage are arbitrary and retrospective (e.g., The loss of one blood volume within a 24-hour period.) Therefore, a practical definition of major haemorrhage adopted by the British Society of Haematology is bleeding which leads to a heart rate of >110 bpm and/or a systolic blood pressure of <90 mmHg.
In children, due to a small total blood volume (80ml/kg), the loss of even small volumes of blood can be significant. Major haemorrhage may be defined as the requirement to transfuse more than 40ml blood/kg.
It may not be straight forward to readily determine that major haemorrhage is occurring, for example post-partum; but early recognition of significant blood loss, ideally before major increments in pulse rate and falls in blood pressure, will allow prompt action to pre-empt shock.
Therapeutic Goals
- To achieve restoration of an adequate blood volume, in order to maintain tissue perfusion and oxygenation.
- Minimise blood loss and achieve haemostasis by:
- Rapid control of bleeding
Damage control surgery and proximal control procedures
Interventional radiology
- Correcting coagulopathy by judicious use of blood component therapy
This major haemorrhage policy should be easily accessible online, in all relevant clinical and laboratory areas. Staff working in these areas must become familiar with them. Deviation from the major haemorrhage policy should be avoided as this can cause confusion, putting the patient at increased risk through delays and an increased likelihood of errors.
Training
Regular training, drills and simulation will improve awareness and confidence and ensure that the major Haemorrhage transfusion process works efficiently.
These need to be carried out regularly due to staff turnover in order to educate and train as many staff as possible. Locations where transfusion is deemed least likely are at the greatest risk of errors. Therefore, training, drills, and simulation should be conducted at all sites where transfusions and major haemorrhage could occur and should include activation of EDP where appropriate.
Communication
There may be more than one major haemorrhage or transfusion event under way that the laboratory is supporting. It is vital that any communications to the lab clearly identifies the name (if available) or unique identifier(s) of the patient and ward or department. In addition, please notify blood bank if / when the patient moves location.
Where possible, advanced warning of a major haemorrhage gives the MTF and blood bank opportunity to prepare. By example, this could include pre-emptive activation of EDP based on 9 liner.
The activation of the major haemorrhage protocol (adult, paediatric or obstetric) will determine how the laboratory staff will respond. On triggering the protocol, the laboratory staff prioritise availability of products.
The auditing of each major haemorrhage event will enable the members of the Defence Medical Services Transfusion Committee (DMSTC) to ensure that it is appropriately triggered. Any wastage of blood or blood product will be investigated as part of the audit.
Clinical team attending the patient
It is for each area to manage these events according to their own requirements, these may be laid down in MTF SOPs, but the following should be considered: -
- Nominate a team lead for each event.
- Nominate one person to make and take all phone calls to Blood Bank during the event.
- Designate a SUITABLY TRAINED individual (usually a porter) to transport samples to the lab and to collect product from the labs. This individual must be trained and competent in use of the electronic blood tracking system.


Protocol activation
Delays in activation can adversely affect patient outcomes, ‘over-activation’ is preferred to ‘under-activation’. All members of the healthcare team should be empowered to activate the major haemorrhage protocol if there is clinical concern.
There is a clear and unambiguous phrase that activates the major haemorrhage protocol. When communicating with blood bank you must follow the protocol activation as described in the section below:
- Notify Blood Bank
The contact details for the duty BMS and/or blood bank should be clearly displayed in clinical areas and inserted into any local copies of MHP.
- Say to the laboratory, “I want to activate the Major haemorrhage protocol”.
For obstetric: “I want to activate the Obstetric Major haemorrhage protocol”.
For paediatric: “I want to activate the Paediatric Major haemorrhage protocol”.
- You will be asked to state your location and patient name or unique identifier (if known), sex (to identify patients of childbearing potential) and approximate age. You will also be requested to provide the name and contact number of your team co-ordinator for the duration of this activation of the protocol.
Any future communication during this time should be preceded by, “this call relates to the major haemorrhage for ………….(state patient name / identifier) in …………. (State clinical area)
Patient Identification
Patient details can be taken over the phone by the blood bank laboratory staff. However, the request form and appropriately labelled sample is required before group specific products are issued.
Except for Emergency O Neg and Emergency O Pos (where applicable) RBC’s Emergency A Platelets and Emergency A FFP, all other issues require the blood bank to know the full patient ID (Four points of Patient identification).
Four points of patient identification (forename, surname, date of birth and unique identification number) are required for provision of group specific products (providing there are sufficient tested samples to confirm patients’ blood group)
Unidentified and Hospital Trauma Patients
The MTF should have an ‘unidentified patients’ policy' which allows the Emergency Department an immediate and effective means with which to safely identify a patient against which product can be issued. This must have a minimum of four identifiers.
NOTE: Once the Unidentified Patient Policy has been used, the UNKNOWN PATIENT ID MUST CONTINUE TO BE USED for the proceeding 24 hours, even if the patient’s actual identity becomes known.
If blood results and/or blood product are issued against the Unknown Patient ID and then the actual patient ID starts to be used, all samples must be repeated before any more blood product can be issued.
The Unidentified Patient ID and the actual patient ID will be seen as separate patients until records can be formally merged by the BMS through liaison with MTF management team.
Patient samples
Get it right first time.
Blood samples need to be labelled correctly to prevent delays in treatment. For patients to receive crossmatched blood, the blood bank requires two samples that have been labelled independently of each other. Accurate patient identification is fundamental to patient safety (SHOT 2021).
Miss-labelling of samples prevents the blood bank laboratory from being able to provide the products required. Having to resample a patient and providing the correctly completed documentation will lead to delays in the patient receiving group specific blood products (not emergency blood).
Consent
Each major haemorrhage event is different, but we must be aware that consent must be sought where possible. In the event the consent is not possible, retrospective consent should be sought at earliest opportunity.
All patients should be made aware transfusion has taken place during their treatment. If post transfusion surveillance / monitoring is required clear details should be provided. If clarity is required, the deployed BMS and/or Centre of Defence Pathology Operational Support can be contacted for advice.
Patients who do not accept or refuse transfusions
Every patient has a right to be treated with respect, and staff must be sensitive to their individual needs as well as acknowledging their values, beliefs, and cultural background.
The priority for a patient undergoing a major haemorrhage is to treat appropriately.
Jehovah’s Witnesses may carry an Advance Medical Directive which directs that no allogeneic blood be administered under any circumstances. In carrying this legal directive Witnesses are simply choosing non-blood medical treatment, and their choice must be respected. It is important that once this has been declared or discovered that it is acted upon and not ignored.
Clinical practitioners must be aware of a Jehovah’s Witness patients’ beliefs in relation to receiving blood or blood products and of the non-blood, medical alternatives to transfusion that may be applicable.
It is essential that any agreement to preserve total clinical confidentiality is strictly honoured.
Venous Thromboprophylaxis
Given the risk of thromboembolic complications, the use of pro-coagulant measures should be ceased once haemostasis has been achieved.
Standard venous thromboprophylaxis should be commenced as soon as possible after bleeding has been controlled, as patients rapidly develop a prothrombotic state in the 24 hours following resuscitation.
Laboratory Investigations
Patient management should be guided by laboratory results and near patient testing (POC) but led by clinical status.
When dealing with an evolving process it is important to reassess haemostatic parameters at least hourly, and after each therapeutic intervention, to monitor the need for and the efficacy of component therapy.
Although it is important to monitor laboratory test results frequently during massive transfusion, the administration of blood/components should not wait for these tests.
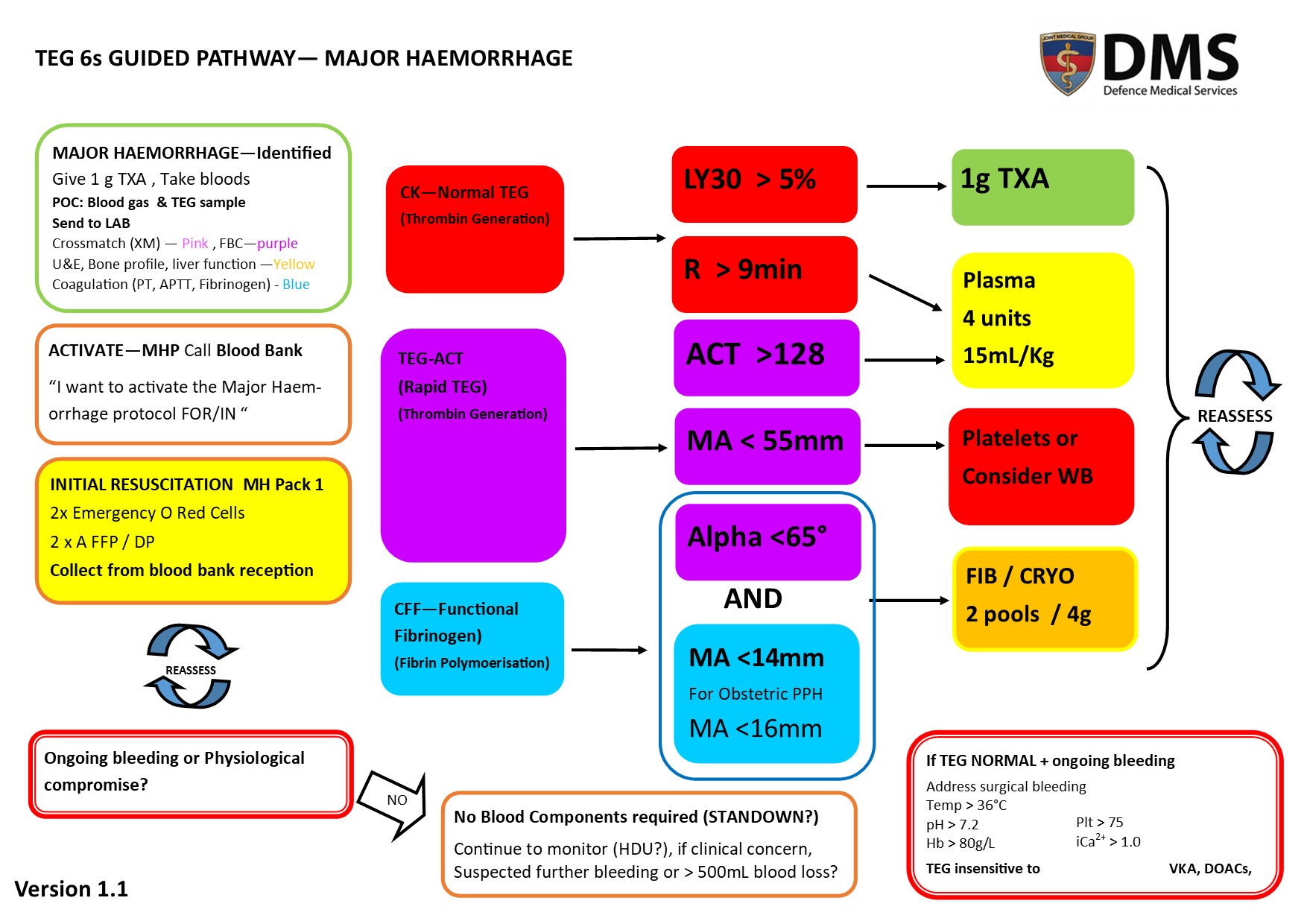
Appropriate use of TEG can offer rapid data to guide component therapy but requires expert interpretation. The TEG CGO algorithms should be followed. It is important to inform Blood Bank if TEG is being employed to direct therapy. Good communication will help blood bank prepare components and reduce delays.
Interpretation of laboratory results
Clauss fibrinogen measurement should be specifically requested during massive haemorrhage situation.
The fibrinogen level is more sensitive than the PT and APTT to a developing dilutional or consumptive coagulopathy. Levels below 1g/l, in the context of massive haemorrhage, are usually insufficient, and emerging evidence suggests that a level above 1.5g/l is required. In Obstetric haemorrhage a level of >2g/L is required. Higher levels are likely to improve haemostasis further. The normal range is between 1.5 and 4.5g/l.
A platelet count below 50 x 109/l is strongly associated with haemostatic compromise and microvascular bleeding in a patient with massive haemorrhage. A minimum target platelet count of 75 x 109/l is appropriate in this clinical situation.
An APTT >1.5 times normal represents established haemostatic failure and is predictive of microvascular bleeding. Early infusion of FFP should be used to prevent this occurring if a senior clinician anticipates a massive haemorrhage. However, correcting to 1.5 times normal is not necessarily an appropriate strategy because haemostatic failure may already be significant at this level. The APTT should be maintained below 1.5 time’s normal as the minimum target.
The PT is an insensitive test for haemostatic compromise and a relatively normal result should not necessarily reassure the clinician. It is common practice to correct the PT ratio to within 1.5 of normal; however, this may not be an appropriate target in many situations.
Management of haemostasis
The indication for use of FFP in massive transfusion with significant bleeding is a fibrinogen <1.5 g/l or PT/APTT >1.5 above normal.
In the context of massive haemorrhage, patients with widespread microvascular oozing or coagulation tests that demonstrate inadequate haemostasis should be given FFP in doses likely to correct the coagulation factor deficiencies. This will require more than 15ml/kg, and at least 30 ml/kg would be a reasonable first-line response.
A minimum target platelet count of 75 x 109/l is appropriate in massive haemorrhage, to provide a margin of safety to ensure that the level does not fall below that critical for haemostasis.
A higher platelet target level of 100 x 109/l is recommended for patients with high velocity trauma, eye or central nervous system injury.
The platelet count should be checked 10-15 minutes after platelet infusion to ensure the adequacy of therapy. A poor platelet increment of less than 20 x 109/l after 15 minutes may be indicative of antiplatelet antibodies, usually human leucocyte antigen (HLA) antibodies.
Cryoprecipitate or Fibrinogen Concentrate should be considered, if fibrinogen levels remain critically low despite administration of FFP or if fluid overload is a concern, and the patient is bleeding.
Fibrinogen Concentrate should be administered in Obstetric Haemorrhage cases where the Fibrinogen level is <2.0g/l
Cryoprecipitate should be administered in traumatic and general Haemorrhage cases where the Fibrinogen level is <1.5g/l
A typical adult dose of cryoprecipitate is two five-donor pools (equivalent to 10 single donor units), containing 3-6 g fibrinogen in a volume of 200 – 500 ml.
A dose (two five donor pools) of cryoprecipitate or 5g fibrinogen concentrate administered to an adult would typically raise the plasma fibrinogen by approximately 1g/l.
Blood Components
Emergency Red Cells (packed red cells or whole blood)
Group O RhD negative is the blood group of choice for transfusion of red cells in an emergency where the clinical need is immediate and patient blood group is unknown.
However, overdependence on group O RhD negative red cells may have an adverse impact on blood stock management.
It is acceptable to give group O Rh D positive red cells to males and females of non-childbearing potential (> 50-year-old) of unknown blood group.
Females of childbearing potential <50-year-old), whose blood group is unknown, should be given group O Rh D negative red cells in order to avoid sensitisation and the risk of haemolytic disease in the newborn.
If Rh D negative components are not available for a woman childbearing potential, transfusion SHOULD NOT be delayed due to concerns of immune sensitisation. The risk of delayed transfusion vastly outweighs risk of immune sensitisation and the priority is to save life.
Women who are Rh D negative and of childbearing potential, whom are resuscitated with Rh D positive blood or platelets, can develop immune anti-D, which may cause haemolytic disease of the new-born in subsequent pregnancies. To prevent this, a combination of exchange transfusion and anti-D can be administered, on the advice of a haematologist, within 72 hours of the transfusion. However, this is unlikely to be feasible on Operations.
Group Specific Blood (BMS required)
In the emergency, blood can be issued following identification of group without knowing the result of an antibody screen – ‘group specific blood’. Blood group determination takes less than 10 minutes, so it should not be necessary to give large volumes of emergency group O blood.
“Group specific Blood” can only be considered when 2 separate samples are tested to confirm the patients ABO group. One of these samples maybe historic, if the patient has a blood group history with blood bank. Patients with no blood group history will require two group and screen samples (Taken and labelled individually of each other, by two separate clinicians)
The use of group specific blood is a higher risk strategy and depends on the urgency for blood. In major haemorrhage, patients will have minimal circulating antibodies, so will usually accept group specific blood without reaction. Antibodies may develop at a later stage.
Emergency Donor Panel activation for Fresh Whole Blood.
Fresh Whole Blood can be collected from EDP donors for a known patient. The SOPs pertaining to EDP activation (0902A – Pre-hospital, 0902B BMS) are held by BMS and/or CD Path and should be followed.
Platelets
1 adult therapeutic dose (ATD) of platelets (either 1 platelet apheresis concentrate or 6 pooled buffy coats) will increase the platelet count by 50 x 109/l
Specific platelet giving sets should be used where possible. Transfusion of platelets through a giving set previously used for red cells is not recommended, as it may cause the platelets to stick to the red cells and therefore reduce the effective transfused platelet dose.
In scenarios where platelets are thought to be clinically required but not available, the use of fresh whole blood (as a source of platelets) via activation of the EDP may be considered.
Plasma (FFP or Dried Plasma)
Early treatment with thawed FFP is recommended in patients with massive bleeding.
FFP is stored at -37oC and it takes 30 minutes to defrost FFP ready for use.
It is crucial that the Blood Bank is informed as soon as possible of a major haemorrhage, so that this defrosting process can begin.
Dried plasma (LyoPlas, FLyP) can be used as a direct replacement for FFP where available. Reconstitution time will be shorter than that required for thawing FFP. Users should be familiar with the process for reconstitution.
Fibrinogen replacement (Cryoprecipitate and Fibrinogen concentrate)
Cryoprecipitate is a concentrated source of fibrinogen and contains factor VIII, factor XIII and von Willebrand factor.
Although ABO blood group compatibility is not required with cryoprecipitate, it is preferred because of the 10-20 mls of plasma in each unit.
Fibrinogen concentrate is dosed in grams and users should be familiar with the process for reconstitution.
In order to increase plasma fibrinogen levels by 1g/L in a 70kg male, a dose of 5g fibrinogen concentrate or 2 pools of cryoprecipitate is suggested.
Blood Storage
Cold chain requirements have become UK law under BSQR 2005.
Red blood cells and FFP should be transfused within 4 hours of leaving a blood fridge.
Red blood cells and FFP cannot be returned to the blood fridge if it has been out of temperature control for more than 30 minutes. In certain situations, blood can be issued in temperature-controlled boxes to extend time out of a fridge, advanced discussion with blood bank is required.
Any product that is not immediately required for use, should be returned to the correct storage location (Blood bank or blood fridge) as soon as possible. This minimises the risk of wastage.
Platelets: A transfusion of platelets should be commenced as soon as possible after they are received. If there is any delay in transfusion, platelets should be returned to blood bank. Platelets must not be refrigerated.
Cryoprecipitate: Must be transfused within 4 hours of thawing. Cryoprecipitate must not be refrigerated.
Traceability
It is a statutory requirement that the ‘fate’ of all blood components must be accounted for. These records must be held for 30 years. Staff must record all blood use in the clinical notes.
When emergency blood is used Blood Bank should be informed immediately and details of which patient has received the emergency blood should be returned to Blood Bank.
Pharmacological Agents
Antifibrinolytics
Fibrinolysis is the process whereby established fibrin clot is broken down. This can occur in an accelerated fashion, destabilising effective coagulation in major haemorrhage situations, including multiple traumas, obstetric haemorrhage and major organ surgery, including transplant surgery.
Accelerated fibrinolysis can be identified by laboratory assay of d-dimers or fibrinogen degradation products or using near patient coagulation testing monitors such as TEG or ROTEM.
Tranexamic acid (TXA)
Tranexamic acid inhibits plasminogen activation, and at high concentration inhibits plasmin, which inhibits fibrinolysis. It should be used in clinical situations where increased fibrinolysis can be anticipated, such as, acute severe trauma.
There is strong evidence that the effect of tranexamic acid on death due to bleeding varied according to the time from injury to treatment. Early treatment (<1 hour from injury) significantly reduced the risk of death due to bleeding events. Treatment between 1 – 3 hours also reduced the risk of death due to bleeding.
Therefore, tranexamic acid should be given as early as possible to bleeding patients as recommended in NICE guidelines 2016.
A loading dose of 1g over 10 minutes followed by 1g over 8 hours is recommended in trauma (as per the CRASH-2 protocol). However, emerging evidence suggests that the second, and subsequent doses could be given as a bolus. Giving the second dose of TXA as a bolus may have practical advantages over the eight-hour infusion in the critically ill patient. There are few adverse events or side effects associated with the early use of tranexamic acid in massive haemorrhage. The key is giving it early, certainly within 3 hours.
Repeat doses should be used with caution in patients with renal impairment, as the drug is predominantly excreted unchanged by the kidneys.
It may be considered in bleeding associated with the direct oral anticoagulants such as Rivaroxaban, Apixaban and Edoxaban.
TXA use in Patient’s Treatment for Gastrointestinal Bleeding
The HALT-IT trial, with over 12,000 patients across 164 worldwide centres (including Derriford Hospital), aimed to clarify if there was justification for the use of TXA in the context of GI bleeding. The results were published in June 2020.
The study showed that TXA did not reduce death from GI bleeding and that, in some cases, TXA increased the risk of thromboembolic events (clots in the veins of the legs that can move to the lungs). An increased prevalence of seizures associated with TXA use, was also reported in this patient group.
Use of Rapid infusion devices
Belmont Rapid infusers can be utilised to rapidly administer warmed red blood cells, FFP and fluids in the massive blood loss scenario.
For Paediatric patients less than 20Kg the Belmont rapid infuser should NOT be used, a ‘Christmas tree’ should be used.
The Rapid infusion devices have a dual purpose:
- The ability to infuse fluids, up to a maximum rate of 750mls/minute
Rapid infusion of fluids/RBC and FFP is crucial to restore circulating volume
- Warming the fluid (Please note: The Warming function is only accessible whilst device is connected to AC power)
Haemostasis is strongly influenced by body temperature, and hypothermia is associated with increased risk of severe bleeding.
Platelets and Cryoprecipitate must NOT be infused via these rapid infusion devices.
The Rapid Infusion devices cannot be operated at pressure to infuse fluids through an intraosseous (IO) access needle. However, the rapid infusion device can be used to warm the fluid, before drawing off fluids into a syringe and delivering through the IO access needle.
Operation of the Rapid infusion devices must only be performed by individuals trained and deemed competent to operate the device.
Considerations for Senior Medical Officer / Clinical Director.
- On Operations blood stocks are limited and careful liaison with blood bank / BMS is required to ensure maximum benefit to the expected patient population. Proactive planning of re-supply is essential to maintain MTF capability.
- In extremis rationing of blood stocks may need to be implemented. Consideration should be given to clinical futility, team resources, blood component; availability, clinical efficacy and safety.
- Consider early activation of EDP when demand for a known patient(s) is likely to outstrip stocks.
- Major haemorrhage activation should be integrated into simulation/moulage scenarios in prior to and during deployment. These should include all elements of MH protocol, including EDP activation.
- In well trained teams, the time from EDP activation to first unit of FWB would be expected to be c. 45 mins with c. 15 mins for each subsequent unit.
- CD Pathology Operational Support should be contacted for SME advice or if concerns regarding deviation from SOPs or protocols.
- Blood Planning Factors based on historical data suggest 20% of casualties attending an MTF will require blood transfusion, with an average of eight whole blood equivalents per patient.
